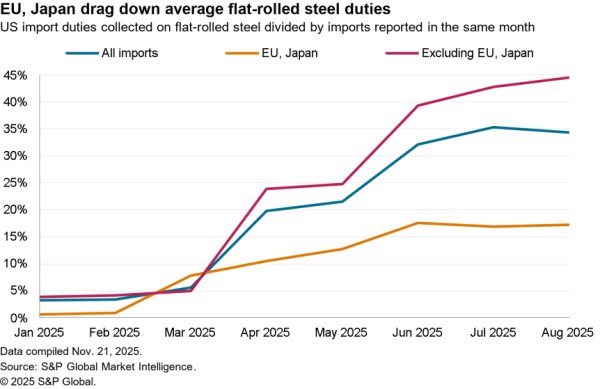
Rising U.S. healthcare costs have led President Donald Trump to call for a wide-ranging review of pricing, including the inclusion of international benchmarks as outlined in Panjiva research of Oct. 29. While that has yet to be converted into firm policy there’s likely to be attention focused on the most expensive classes. Among drug imports the second largest class were cardiovascular drugs – including statins – with $5.26 billion of imports in the 12 months to Aug. 31, Panjiva data shows.
Imports of the drugs covered by the class fell 14.7% on a year earlier including a 7.8% rise in volumes, a potential proxy for number of scripts written, and a 22.5% slide in average import values per gram (AIVG).

Source: Panjiva
The decline in average import costs and rise in scripts may reflect the availability of generic versions of Merck’s Zetia (ezetimibe) whose original patent ended in April 2017. Subsequent to that the share of ezetimibe imports has risen to 11.5% of total statin imports (covering six major generic drug types) from 1.3% a year earlier. The main losers have been pravastatins (22.1% from 32.5%) and atorvastatins (13.0% from 21.2%).

Source: Panjiva
The leading shippers of ezetimibe to the U.S. since August 2017, who are able to profit from the end of Merck’s patent, include Lek Pharma (38.9% of shipments, sourced from Slovenia) and Alkem (27.8%, from India) though Watson, Amneal and Aurobindo also appear to be shipping commercial volumes.

Source: Panjiva